- Home
- Clinical Trials
Basal Cell Carcinoma Studies Auckland
BETHELLS Basal Cell Carcinoma Study
Auckland
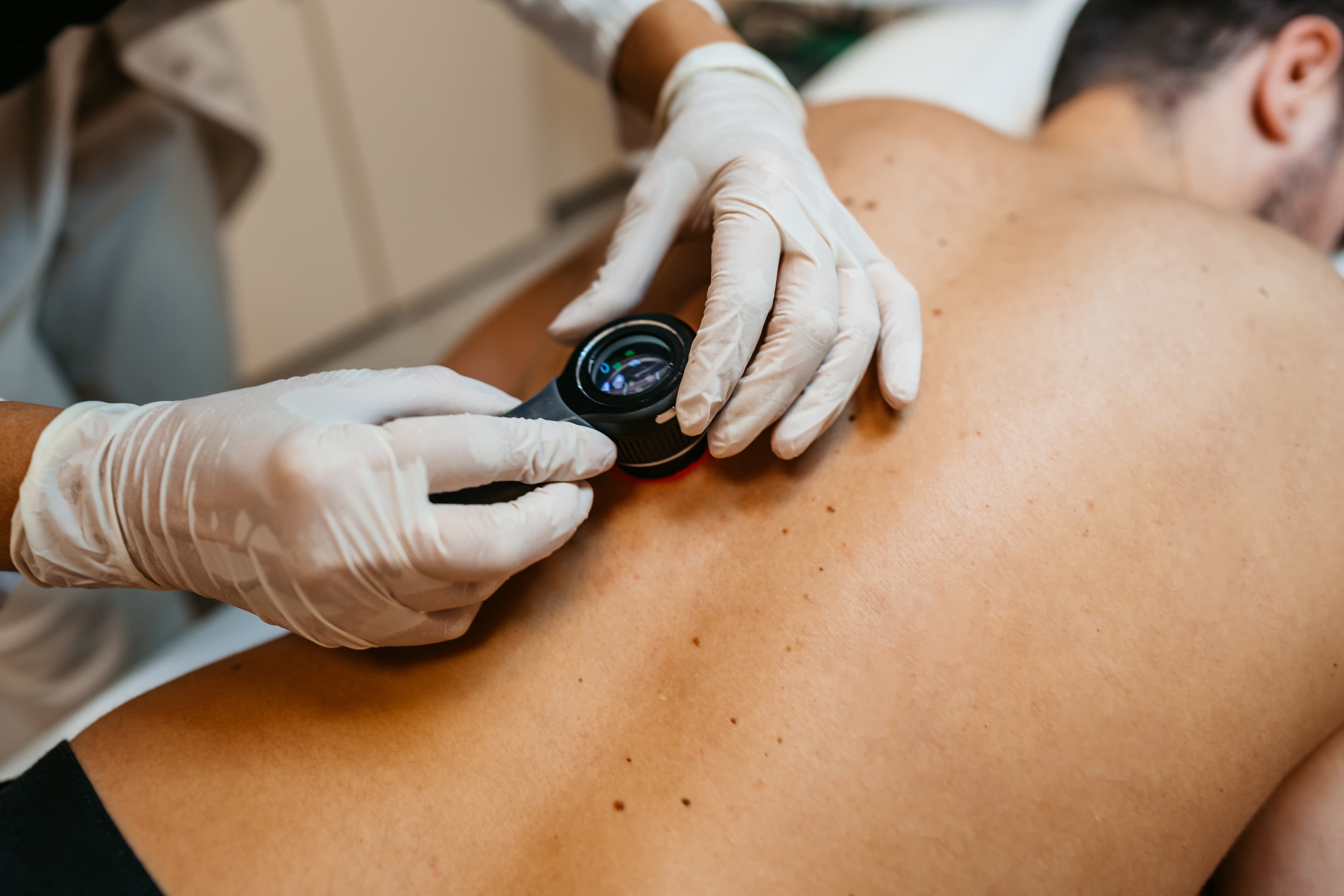
Basal cell carcinoma (BCC) is a common cancer. Nodular BCC (nBCC) is the most common type of BCC. It usually appears as a pearly or flesh-coloured, shiny lump, but can take other forms. It most often forms on the face, neck and other areas exposed to the sun. Although nBCC is the least dangerous type of skin cancer and rarely spreads, it can cause tissue damage if left untreated. These tumours are usually surgically removed.
Description
What to expect
Study details
Ages
18 years or older
Duration
Up to 10 clinic visits over 52 weeks (1 year)
Compensation
You will be reimbursed for your time and travel costs
Eligibility Criteria
- 18+ years with a nodular basal cell carcinoma
- Have light to medium skin tone (required for laser light safety)
- Otherwise, healthy
- Willing to undergo study procedures, including tumour punch biopsies
Locations
Description
Most basal cell carcinomas are treated with surgery, cryosurgery and radiotherapy.
The study product (TSK-001) is a potential new treatment for skin cancers. The study product is injected into your nBCC and then exposed to NIR light to heat and kill the tumour cells. After the injection, you’ll be monitored for about 2 hours to ensure you don’t have any reactions.
What to expect
Your involvement in this study may help advance scientific knowledge that could lead to the development of a new treatment for people with nBCC. Other reasons to participate may include:
- Receive treatment and monitoring of your nBCC by a skin specialist and NZ doctors and nurses
- Your nBCC may clear without the need for surgery.
Taking part in the study involves firstly meeting with the research Doctor to find out more about your health and to see if the study is right for you. If you wish to take part and are eligible, we would like to see you for up to 10 clinic visits over 52 weeks (1 year).
During visits, we conduct health checks, which may include physical examinations.
- Vital sign checks (blood pressure, heart rate, temperature)
- ECGs to monitor heart activity
- Blood, urine and tumour samples
- Questions about medications, adverse events and pain.
About 78 people worldwide are participating in this study.
Apply now
Fill out your details, and our team will be in touch to discuss your potential involvement in this study and answer any questions you may have.
Momentum Clinical Research NZ is a member of the Global Site Holdings Group. Global Site Holdings Pty Limited ACN 668 977 062 trading as Momentum Clinical Research ABN 51 668 977 062 and our related entities (we, us, our), will use the Personal Information you have provided in the above form to review and respond to your enquiry, to contact you about any future trials or studies you may be eligible for, and otherwise in accordance with our Privacy Policy and as required and authorised by law. We may not be able to provide you with a complete response or a response at all to your enquiry if you do not provide the requested information. Our Privacy Policy also contains further information about how we collect, use, store or disclose your Personal Information, how you may seek access and correction of your Personal Information, and how you may make a complaint about a breach of privacy. If you have questions, please contact us at contact@momentumclinicalresearch.com.